Receive free delivery of FCI to your Inbox and a weekly newsletter

Subscribe to FloraCulture International Magazine


The International Association of Horticultural Producers (AIPH) members consist of thousands of growers of flowers and ornamental plants around the world who are together united by one essential goal – promoting the place of plants in people’s lives.
About
Contact
Join AIPH


Our Annual Review in 2023 marks AIPH'S 75th Anniversary and the many activities we have achieved
Learn moreAIPH is responsible for approving the world’s most exceptional horticultural expos creating long lasting green legacies in cities across the globe.
Overview
Media
Partners
Approved Expos
Expo Support Programme
Contact
AIPH Blooming Economies & Cities to Inspire the World


AIPH Blooming Economies & Cities to Inspire The World 2.0. Up-to-date statistics from recent Expos hosted in Asia and Europe.
Learn moreAIPH hosts several conferences, webinars and events such as the International Grower of the Year award throughout the year. Read about these along with the latest news about the association’s activities.
News
Previous Events
Contact
AIPH International Grower of the Year


The AIPH International Grower of the Year Awards celebrate the best of the best in ornamental horticulture
Learn moreAIPH’s key aim is to stimulate demand for ornamental trees, plants and flowers worldwide. We are committed to supporting growers in achieving this by protecting and promoting the interests of the industry.
Overview
Ornamentals Groups
Contact
The AIPH’s Global Industry Intelligence Centre (GIIC) is an international hub for industry information and knowledge exchange.
Statistical Yearbook
Consultancy and Bespoke Reports
Contact
Understanding The Chinese Market
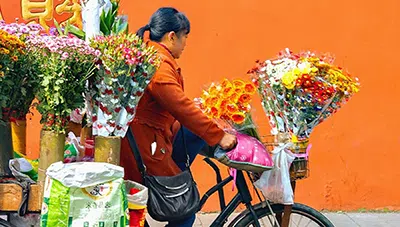
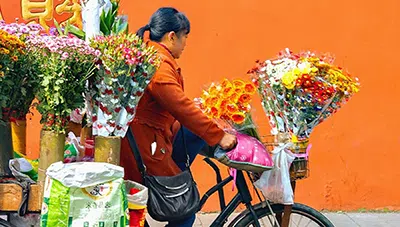
The report contains research into; Working middle class in China. Domestic production in China & Retail research summary of the floral retails survey.
DownloadAIPH Green City activities promote the essential role of plants in creating vibrant urban areas in which people and businesses can thrive.
Overview
- Green City Home
- Conferences
- Committee
- Green City Briefings
- Global Green City Forum
- Living Green, Liveable Cities Research Symposium
News
Green City Guidelines
Green City Awards
Contact